|
Ii) In XRD profiles, the forbidden reflections typically have absolutely zero intensity. Especially, unlike XRD, CBED patterns are very sensitive to the presence or absence of a center of symmetry. For electron diffraction, multiple ( dynamical) scattering of the incident electrons in the TEM specimen occurs so that the Friedel's law is broken down, and thus the crystals with all the 230 space groups can be identified. This limits the number of the space groups of crystals that can be determined by XRD to fifty. I) XRD obeys Friedel's Law because it can be approximately described by kinematical scattering. Then, the difference between the two techniques is mainly on systematically forbidden reflections: More information on the advantages of electron diffraction techniques can also be found in this book.įor symmetry determination with both XRD and electron diffraction crystallography, we are looking for symmetry-related reflections. Viii) A sample that appears crystalline to a local probe such as nano-beam electronĭiffraction may appear amorphous to powder X-ray diffraction because both techniques have very different spatial resolutions.įor instance, the structures of several unknown compounds including ceramic oxides, a new Ti–Se compound and an Al mFe precipitate in an aluminum alloy have been solved with electron diffraction techniques. Vii) Powder x-ray diffraction provides the lattice-plane spacings and the corresponding intensities, but does not give the interplanar angles. Vi) In contrast to X-ray diffraction, the Ewald sphere is very flat for electron diffraction in TEM. V) X-ray diffraction statistically gives us a good idea of the average sample, electron diffraction in TEM allows us to obtain local structure information. Iv) The EM is the only technique that can provide a microscope image as well as a diffraction pattern at atomic resolution. The stronger interaction of the incident electrons with matter allows us to obtain the information about subtle crystallographic details that are usually undetectable in X-ray diffraction technique Iii) X-rays are much less sensitive than electrons to changes in the electrostatic potential. Ii) The atomic scattering factors for electrons are very different from those for X-rays and it is easier for electron diffraction to observe light atoms in the presence of much heavier atoms high T c superconductors, are too small in size and are too imperfect in periodicity for an X-ray single crystal analysis to be performed, but are applicable for electron-microscopic (EM) observations For instance, the grains of many crystalline materials, e.g. Furthermore, higher spatial resolution is needed because the surface and near surface regions of nanocrystals are highly distorted or relaxed with respect to the bulk crystal structure. However, the situation for smaller crystals becomes rather complex and the Bragg peaks may shift simultaneously as well as get asymmetrically or even anisotropically broadened, especially the powder X-ray diffraction for nanocrystals become less and less characteristic because more and more Bragg peaks overlap due to the peak broadening and the intensity diminishes until they become difficult to distinguish from the background. I) The method of powder X-ray diffraction works best for micrometer-sized crystals and becomes less useful for crystals in the nanometer range because the kinematic X-ray diffraction from perfect crystal lattices shows essentially delta functions for the line profiles of the individual reflections. However, electron-crystallographic methods still have their advantages: Comparing with X-ray crystallography, the major difficulties in TEM are smaller tilt range with goniometer stages and smaller physical dimension of specimens placed in the objective lens polepiece gap. 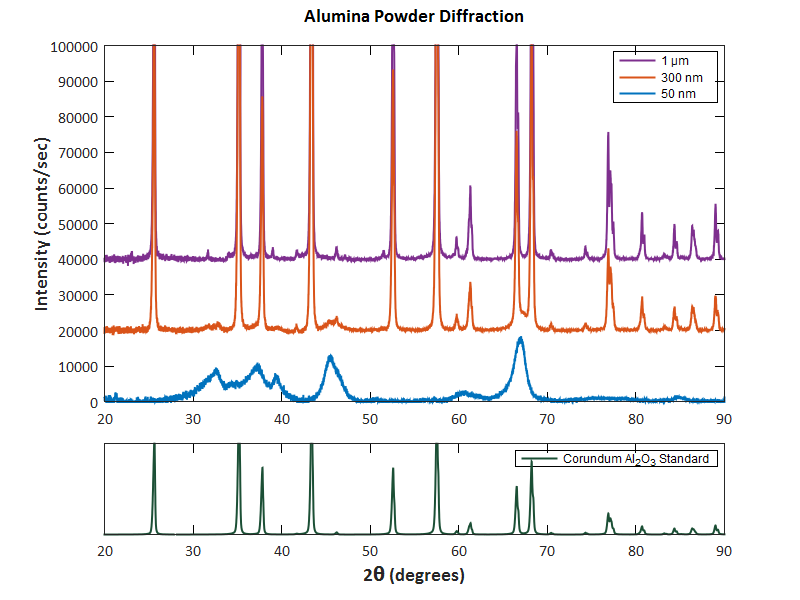
Electron diffraction is usually complementary to powder X-ray diffraction. Since the Russian work as early as 1950s, electron crystallography has been continuously developed as an effective technique for solving the difficult crystal structures, which could not be solved by X-ray methods.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
